Back to Journals » Drug Design, Development and Therapy » Volume 19
The Toxicity of Dimethyl Sulfoxide Combined with the Alkylating Agent MNU on Mice: Focusing on Mortality and Activity Impairment
Authors Yan W, He Q, Xiao L, Xia Y, Zheng X, Zhang X
Received 8 February 2025
Accepted for publication 28 May 2025
Published 5 July 2025 Volume 2025:19 Pages 5727—5737
DOI https://doi.org/10.2147/DDDT.S521506
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Anastasios Lymperopoulos
Weiming Yan,1 Qiurui He,2 Lin Xiao,1 Yuxin Xia,1 Xiangrong Zheng,1 Xiaohong Zhang1
1Fuzong Clinical Medical College of Fujian Medical University, Fuzhou, 350025, People’s Republic of China; 2The Third Hospital of Zhangzhou, Zhangzhou, 363005, People’s Republic of China
Correspondence: Weiming Yan, Email [email protected] Xiaohong Zhang, Email [email protected]
Objective: Dimethyl sulfoxide (DMSO) is known to enhance the absorption of chemicals. This study investigated the effects of DMSO in combination with the alkylating agent, N-methyl-N-nitrosourea (MNU), on the activity and mortality of mice, providing foundational data for solvent mixture use and health impact assessment.
Methods: Twelve male SPF C57BL/6J mice were divided into three groups (n=4): DMSO group (D), DMSO combined with low-dose (L), and high-dose MNU group (H). Mice in D received intraperitoneal injections of pure DMSO solution at 12 mL/kg. The L or D group was injected with a DMSO solution containing 40 mg/kg or 60 mg/kg MNU at 12 mL/kg, respectively. Mice were monitored for changes in body weight, activity levels (walking, body stretching, fur condition), and mortality at various time points.
Results: Pre-injection, all groups showed no statistical differences in weight or DMSO dose, while the MNU injection dose was significantly different (H>L>D). Within 2 hours post-injection, all mice except one in the D group exhibited reduced mobility, hunched posture, and lethargy. Mortality progressed rapidly, with one D and two L mice dying by 12 hours, followed by additional deaths (one D, two L, and two H mice) at 24 hours. By 36 hours, all mice in L and H groups had died, while one D mouse recovered normal activity. At the 48-hour endpoint, only one D mouse survived with normal behavior. No significant differences in weight, activity levels and mortality changes were observed in surviving mice throughout the study period.
Conclusion: DMSO has certain toxicity, and when combined with alkylating agents such as MNU, it can lead to reduced activity and an increased mortality rate in mice. It is recommended to closely monitor the mice during the use of such reagents and to establish appropriate research observation protocols.
Keywords: dimethyl sulfoxide, MNU, toxicity, mice, mortality, physiological effects
Introduction
Dimethyl sulfoxide (DMSO) is a highly polar, organic compound that is widely utilized in scientific and medical researches.1,2 Known for its unique ability to penetrate biological membranes, DMSO is often used as a solvent to facilitate the absorption of a wide variety of chemical compounds, including drugs, toxins, and other substances. This characteristic makes DMSO an invaluable tool in laboratory experiments, particularly in drug delivery systems and pharmacological studies.3 In particular, DMSO’s ability to increase the permeability of biological membranes can influence the pharmacokinetics and biodistribution of co-administered substances, potentially altering their efficacy and safety profiles. This is especially relevant in toxicological studies, where DMSO is frequently used as a vehicle solvent to solubilize poorly water-soluble compounds. However, its capacity to enhance the absorption of compounds into biological tissues also raises concerns about its potential to exacerbate the toxic effects of those compounds, as high concentrations or prolonged exposure may lead to adverse effects such as hemolysis, allergic reactions, and genotoxicity.
One of the critical areas where DMSO’s solvent properties are frequently leveraged is in the use of chemical agents such as alkylating agents, which have been extensively studied for their ability to modify the structure and function of biological molecules, particularly DNA. Alkylating agents, including N-methyl-N-nitrosourea (MNU), one of the most commonly used alkylating agents, has been widely utilized in cancer research and retinitis pigmentosa modeling.4,5 These agents could covalently bind to DNA, leading to significant alterations in its structure and function. Specifically, they exert their effects by transferring alkyl groups to DNA, which can cause mutations, chromosomal damage, and eventually carcinogenesis or apoptosis of retinal photoreceptors. MNU, in particular, is a powerful mutagen and carcinogen that has been shown to induce a variety of cancers in laboratory animals, including lymphomas, leukemias, and solid tumors in various organs. Its high reactivity and ability to penetrate biological membranes make it an effective agent for inducing genetic damage in a wide range of cell types. Besides, the effectiveness of MNU as a research tool for studying retinitis pigmentosa has made it a cornerstone in many ocular experimental protocols.6,7 In details, by selectively targeting retinal photoreceptors, MNU allows researchers to mimic the progressive degeneration seen in human retinitis pigmentosa, providing a valuable model for testing potential therapeutic interventions.
While DMSO is often regarded as relatively safe when used at low concentrations, its interaction with other chemicals and the subsequent effects on the health of experimental animals require careful consideration. Low levels of DMSO may interact with cellular processes or metabolic pathways in ways that are not yet fully understood. Studies involving DMSO as a vehicle solvent for chemical agents need to take into account the potential for DMSO to alter the pharmacokinetics and toxicity profiles of the compounds being studied. It may influence absorption rates, distribution within tissues, and elimination half-lives, potentially leading to unexpected outcomes. For example, DMSO was shown to increase the permeability of the blood-brain barrier and enhance the systemic distribution,8 which could result in higher concentrations reaching sensitive areas such as the central nervous system, thereby amplifying their therapeutic or toxic effects. However, despite the well-documented uses of DMSO and MNU in research,9 the combined effects of these substances on biological systems have not been fully elucidated, particularly in terms of long-term consequences or subtle interactions that may only become apparent after prolonged exposure. The potential of DMSO to enhance the absorption of MNU into biological tissues might lead to a more pronounced toxicological effect than when either agent is administered alone. This could result in increased DNA damage or oxidative stress, which are critical factors in carcinogenesis and tissue injury. Given that alkylating agents like MNU can induce significant DNA damage, it is important to assess the overall toxicity and safety of these compounds of DSMO and MNU, particularly in animal studies, to ensure that experimental protocols adequately account for potential risks and to establish safe dosage ranges for future research applications.
The main goal of this study was to investigate the combined toxicity of DMSO and MNU in a controlled animal model. Specifically, this research aimed to assess the impact of DMSO on the toxicity and mortality associated with MNU exposure in C57BL/6J mice, which are commonly used in ocular researches. By varying the doses of MNU and administering it in combination with DMSO, this study provided preliminary data on the synergistic effects of these two compounds. Additionally, the study assessed whether DMSO, by facilitating the absorption of MNU, exacerbated the toxicological outcomes, including changes in activity levels, health status, and overall survival rates in the mice.
Methods
Animal Models
A total of 12 male SPF C57BL/6J mice, aged 8–10 weeks, with an averaged body weight range of 20–25 grams, were selected for this experiment. These mice were obtained from the Laboratory Animal Center of Fuzong Clinical Medical College of Fujian Medical University (License number.: SYXK2018006) and were allowed to acclimate to the laboratory environment for a period of 3 days before the commencement of the study. The mice were housed in standard laboratory conditions, with a controlled room temperature of 22°C ± 2°C and a relative humidity of 50–60%. The animals were maintained on a 12-hour light/dark cycle, with lights on at 7:00 AM and off at 7:00 PM. The mice were provided with standard rodent chow and clean, filtered water ad libitum throughout the experiment. The cages were kept in an environmentally controlled room to minimize external stressors, and the mice were allowed unrestricted access to their environment in order to reduce potential confounding factors.
After a period of 3 days of accommodation, the mice were randomly assigned to one of three experimental groups, with four mice per group. The groups were: the DMSO control group (D), the low-dose MNU group (L), and the high-dose MNU group (H). We confirmed that ethical and legal approval was obtained from the Experimental Animal Care and Use Committee of Fuzong Clinical Medical College of Fujian Medical University (Ethics committee number: 201923), prior to the commencement of the study. Besides, all animal experiments and procedures were performed in accordance with the ethical guidelines and regulations established by Fuzong Clinical Medical College of Fujian Medical University and our nation. The overall design of our study was showed in the Graphic Abstract of the Study.
Group Design and Treatment Protocol
- DMSO Control Group (D): The D group, serving as the vehicle control, received intraperitoneal (IP) injections of pure DMSO (100% concentration). Each mouse was administered 12 mL of DMSO per kilogram of body weight. This control treatment was designed to assess the baseline effects of DMSO, ensuring that any observed effects in the other groups could be attributed specifically to the MNU treatment, rather than the vehicle itself.
- Low-dose MNU Group (L): The L group received a DMSO solution containing 40 mg/kg of MNU,10 the potent genotoxic agent. MNU was dissolved in DMSO to obtain the desired concentration, and the injection volume for all mice was maintained at 12 mL/kg body weight. In greater details, we firstly calculated the required amount of MNU. For example, for a dose of 40 mg/kg in 3 mice weighing 30 g each, the amount of MNU needed would be MNU = 40×10 × 0.03 = 12 mg. Then, we calculated the corresponding concentration of the MNU solution for injection at a volume of 12 mL/kg. If the MNU was administered at 40 mg/kg and injected at a volume of 12 mL/kg, the corresponding concentration of the MNU solution should be 10/3 mg/mL. Thus, 12 mg of MNU would require a volume of DMSO of 3.6 mL. The solution was then stored at 4°C for later use, and prepared immediately before use. The purpose of this group was to examine the effects of a low-dose MNU exposure on the mice, focusing on toxicological effects and any changes in behavior or general health that could result from this exposure level.
- High-dose MNU Group (H): The H group received a higher dose of MNU, specifically 60 mg/kg,10 in the DMSO solution. Similar to the L group, the MNU solution was prepared by dissolving MNU in DMSO, and the injection volume was 12 mL/kg of body weight. This higher dose aimed to assess the potential acute toxicity and mortality associated with a more severe exposure to MNU. This group was expected to show more pronounced toxicological effects, including possible lethality, and served as an extreme comparison for the lower dose.
Injection Procedure
All injections were administered via intraperitoneal injection, a standard route for delivering substances that need to be absorbed into the body quickly. The injections were carefully carried out by an experienced personnel to ensure precise and consistent dosages across all animals. The animals were weighed immediately prior to injection to ensure accurate dosing, and each mouse was handled gently to reduce stress and minimize discomfort.
Monitoring and Observations
Following treatment, the mice were closely monitored for any immediate or delayed signs of distress, toxicity, or adverse effects. The following parameters were tracked throughout the study:
- Body Weight: Mice were weighed several timepoints (Timepoint 1 (T1): at the time of purchase (ie, before the 3-day pre-adaptation feeding); T2: before injection (ie, after the 3-day pre-adaptation feeding); T3: at 24 hours post-injection; T4: at 48 hours post-injection) to monitor any significant changes in body weight, which could indicate adverse effects such as dehydration, malnutrition, or systemic toxicity. A marked decrease in body weight was considered a key indicator of toxicity,11,12 and the rate of weight loss was recorded for comparison across groups.
- Activity Levels and scoring: Activity levels were observed both during the light and dark cycles at several timepoints (T1: before injection; T2: at 2 hours post-injection; T3: at 24 hours post-injection; T4: at 36 hours post-injection; T5: at 48 hours post-injection). General locomotor activity was evaluated through simple observational methods, such as monitoring the frequency of walking, movement across the cage, and exploratory behavior. Mice were also monitored for signs of lethargy or reduced activity, which are common indicators of stress or illness.13 Additionally, behaviors like body stretching and body posture were noted, as these could reflect changes in the mice’s physical condition or discomfort.14,15 Besides, fur condition was examined daily to assess any signs of stress or health decline. Deterioration in fur quality, such as loss of fur, roughness (or called the ruffled fur), or lack of grooming behavior, was considered an important indicator of poor health or systemic effects of the treatment.15,16 In our study, the activity score was primarily based on three key domains: the mice’s activity alertness (or the locomotor activity), body extension or posture, and the condition of the fur. A total of 9 points were assigned, with 3 points allocated to each domain. The corresponding scores were assessed and recorded by one certain person, and then were stored for later statistical analysis.
- Mortality and Survival curve: Mortality was monitored at several timepoints (T1: before injection; T2: at 2 hours post-injection; T3: at 24 hours post-injection; T4: at 36 hours post-injection; T5: at 48 hours post-injection). Any deaths within this period were carefully observed and the number of mice in each group were recorded. Furthermore, the surviving time of mice in each group was recorded and the survival curves were created based on the number of surviving mice and their survival time.17 Further Kaplan-Meier survival curve analysis was then performed.18
Statistical Analysis
Due to the small sample size of 4 animals (less than 5 animals) per group, the non-parametric statistical method, the Bootstrap test, was employed to compare the groups.19 This special test was chosen as it does not rely on distributional assumptions, making it more appropriate for small sample sizes.20 The data were presented as mean ± standard deviation. In the Bootstrap method, the calculation of the P-value is based on the distribution of resampled data and does not directly provide a standard P-value. Although there is no direct P-value, the significance can be indirectly assessed through the following approach by examining the confidence interval. If the confidence interval of the Bootstrap results does not include zero, this typically indicates that the difference is significant. The survival analysis was conducted using the Kaplan-Meier survival curves. The statistical analysis was performed using SPSS 27.0 software, and survival curves were generated and analyzed using Log-rank tests. Figures were generated using GraphPad 5.01 software.
Results
Pre-Injection Status and Parameters
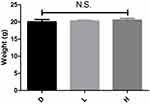
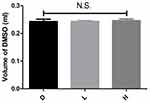
Before the injection, the mice in all experimental groups were closely monitored. The 95% confidence interval of difference in the body weight of mice revealed by the Bootstrap analysis between the D and L, D and H, L and H groups, were (−3.16, 2.66), (−3.36, 2.36) and (−2.25, 1.75) respectively. No significant statistical differences were found in terms of body weight, as the confidence interval of the Bootstrap results included zero (Figure 1). The 95% confidence interval of difference in the volume of DMSO injected between the D and L, D and H, L and H groups, were (−0.019,0.012), (−0.025, 0.012) and (−0.016, 0.007) respectively. No significant statistical differences were found as the confidence interval of the Bootstrap results included zero (Figure 2). It suggested that the experimental groups were well-matched at baseline and there was no noticeable variation in these parameters before treatment.
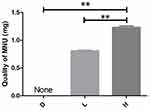
Regarding the MNU injection, the dose administered to the mice in the H group was significantly higher than that given to L group (the 95% confidence interval of difference was 0.076–0.096), which was in turn higher than the dose given to the D group (the 95% confidence interval of difference was 0.160–0.166). The statistical analysis confirmed that the differences in MNU doses among the groups were highly significant, as the confidence interval of the Bootstrap results did not include zero (Figure 3). Prior to the injection, all mice in the study exhibited strong mobility, alertness, and a relaxed posture, with their fur appearing glossy and well-groomed, indicating overall good health.
Survival Counts and Survival Curves Analysis
Following the injection, the survival rates varied across the different groups. Two hours after the injection, all mice in all the experimental groups, except for one mouse in the D group, displayed reduced mobility, decreased alertness, and a hunched posture. After 12 hours, mortality of the mice in all groups began to increase, with one mouse in the D group and two mice in the L group being dead. By 24 hours, the mortality rate continued to rise, as another one mouse in the D group, and two mice in both the L and H groups died. After 36 hours, another one mouse in the D group had died, while one mouse in the D group had regained increased activity. The other mice exhibited the poor mobility and the hunched posture. At this point, all mice in the L and H groups had died, leaving only two surviving individuals in D group. After 48 hours, the recovering mouse in the D group showed significant differences in the survival, which remained alive, exhibiting normal walking ability and alertness. Meanwhile the other mice in the D group had died out (Figure 4).
Body Weight Changes
Throughout the course of the study, no significant differences in the changes of body weight were found in the surviving mice across all groups in all the timepoints of observation. Despite the varying mortality rates and changes in activity levels among all groups, the body weight of the surviving mice remained stable in the D, L and H groups. This might suggest that the physiological effects of the treatment did not result in substantial weight loss among the survivors during the observation period (Figure 5).
Activity Levels Changes
In terms of activity, two hours after the injection, all mice in the experimental groups showed a marked decline in activity levels. They appeared less alert, displayed a hunched posture, and had reduced physical activity compared to their pre-injection status. As the hours passed, the decline in activity became more pronounced. By 24 hours, some mice in all the groups exhibited trembling, and their activity levels were further reduced. At 36 hours, one mouse in the D group managed to regain increased activity, while others showed signs of poor mobility and distress. No significant differences in activity levels scores were found in the surviving mice across all groups. By the 48-hour mark, the only surviving mouse in the D group displayed the normal walking ability and alertness, while all the other mice in the D, L, and H groups had died out (Figures 6 and 7).
Discussion
In our study, male C57BL/6J mice were divided into three groups, the D, L and H groups. Mice were monitored for changes in body weight, activity levels, and mortality. Our results showed no significant differences in body weight among the groups prior to injection. Mortality rate was relative increased with injection of DMSO and MNU combination, while the DMSO alone could also led to a high mortality. All mice in the L and H groups had died out by 36 hours, while only one mouse in the D group survived, regaining a normal activity by 48 hours. All mice exhibited a reduced mobility and a hunched posture, with the activity declining progressively, after injection of either the DMSO or the DMSO-MNU combination.
In greater details, the mice with only the DMSO injection showed a noticeable decline in activity, reduced alertness, and signs of physical distress such as, a hunched posture, the limb curling and dull fur within two hours after injection. These symptoms indicated the presence of toxic effects. Subsequently, the animal deaths were observed, with only one mouse managed to recover and regain normal vitality at the end. The above results suggested that DMSO had a clear toxic effect on animals, with the potential for lethality or mortality. The toxicity of DMSO observed in this study were consistent with findings reported in other researches.21 DMSO was shown to provoke toxic reactions in the body, which might include a variety of adverse effects.22 Several studies also highlighted the toxicological risks associated with DMSO, indicating that it could significantly alter physiological processes and lead to behavioral changes.23 In particular, the impact of 0.1–10% DMSO concentrations on behavior of aquatic model species has also been noted.24 The DMSO concentrations under 1% did not cause statistically significant mortality, but did induce clear signs of stress, reduced locomotion, and impaired responses to stimuli. Given all these observed effects, it is essential for researchers to be cautious when using DMSO in experimental settings, ensuring that proper controls and safety protocols are in place to safeguard the well-being of animal subjects. It is critical that appropriate dilution of DMSO concentrations is conducted in related studies to minimize potential harm. Interestingly, the observation that one mouse with only the DMSO injection showed activity recovery at 36 hours post-injection was noteworthy. While the DMSO solution itself at high concentration is generally considered toxic, the occurrence of such a recovery in an individual animal may point to inherent variability in the response of mice to the DMSO. It was plausible that the special mouse had a stronger immune or repair response, which allowed it to gradually recover from the toxic effects of the DMSO injection. However, this singular recovery did not diminish the overall trend of severe toxicity observed in the DMSO group, which highlighted the potent and rapid toxicity of DMSO exposure.25
In mice with injection of the DMSO and MNU combination, the DMSO and MNU combination did not exhibit significantly aggravated toxic reactions or increased mortality in mice during the early stages. From the survival curves, it was apparent that there were no noticeable differences in the early stages compared to the DMSO injection. However, as time progressed, the mortality rate in the DMSO and MNU combination groups began to increase significantly. Upon further analysis, it was found that in the DMSO and MNU combination groups with different doses of MNU, there was no clear MNU-dose dependent increase in toxicity at the early stages. Surprisingly, the early mortality rate in the L group was slightly higher compared to H group. As the observation period extended, however, the mortality rates between the two groups became similar. The results might suggest that the relationship between the MNU dosage and the related toxicity may not always follow a straightforward, dose-dependent pattern. It implied that other factors, such as the rate of absorption, metabolism, or individual genetic responses, might influence the observed outcomes of possible toxicity. The early increased mortality in the L group might be attributed to a higher sensitivity of certain physiological systems or an accelerated onset of toxicity in those mice. As time went on, however, the toxic effects of MNU probably reached a threshold, and the overall mortality rate stabilized in both L and H groups.
MNU was initially recognized as a potent carcinogenic agent and has been widely used to induce various types of cancer in animal models.26 Subsequent studies demonstrated that MNU at the dose 60 mg/kg could selectively induce apoptosis in retinal photoreceptor cells, which led to its use in research on retinal pigment degeneration and retinal degeneration-related diseases.7,27 However, there has been limited researches on the effects of MNU in increasing animal mortality and lethality. Some studies reported that higher doses of MNU (80, 160 or 240 mg/kg) could lead to tumor formation, leukemia, and even an increase in early mortality in mice26,28. MNU exposure was showed to impair hippocampal neurogenesis in rats,28 which might lead to toxic behaviors. Our study utilized a relatively low dose of MNU, ranging from 40 to 60 mg/kg, and the results suggested that MNU could, to some extent, enhance the toxic effects of DMSO in mice. This enhancement may be due to an increased inherent toxicity of MNU itself, or a result of MNU-mediated potentiation of the DMSO toxicity.
Several limitations existed in our study. One limitation of this study was the relatively small sample size, which may reduce the statistical power and generalizability of the findings. As our research involved potentially severe toxicological effects and mortality endpoints of mice, we adhered to the principle of minimizing animal use while maintaining scientific validity. This approach aligned with the 3Rs (Replacement, Reduction, and Refinement) principle in animal research. Although we applied the Bootstrap method due to the small sample size, the results should be interpreted with caution and considered as preliminary observations that provided the foundation for more robust studies. Additionally, the short observation period of 48 hours post-injection in our study did not account for potential long-term toxic effects or delayed pathological changes, limiting our understanding of DMSO and MNUinduced chronic toxicity. Besides, the current study did not consider the impact of gender and age variations, which could influence the results. Furthermore, while the study focused on acute toxicity, it did not explore the underlying molecular mechanisms, such as DNA damage repair or oxidative stress. In addition, we did harvest the blood or organs for future enzymatic assays, which might extract the maximum amount of information on the toxicity mechanism. Finally, the use of mice as model organisms, though standard, might not fully represent the human responses. This limitation underscored the necessity for future studies to incorporate other animal models, cell models or human data for broader applicability.
In conclusion, our study demonstrated that DMSO exhibited a notable toxicity, particularly when used in combination with MNU, the alkylating agents. This combination was shown to significantly reduce the activity levels and increase the mortality rate in mice. These findings highlighted the importance of closely monitoring animal’ reactions during experiments involving DMSO and similar reagents. To mitigate potential adverse effects, it is crucial to establish comprehensive research observation protocols that ensure the health and well-being of the animals. Further studies are needed to explore the underlying mechanisms of DMSO and MNU toxicity and to determine the safest concentrations and guidelines for its application in scientific research. Besides, we would include enzymatic assays of relevant serum and organs, including the heart, liver, spleen, lungs, and kidneys of mice, to further explore the possible toxicity mechanism. By advancing the understanding of DMSO and MNU toxicity, it could better inform safety practices and contribute to the broader field of chemical toxicity assessment.
Data Sharing Statement
The datasets used and/or analyzed during the current study were available from the corresponding author on reasonable request.
Acknowledgments
We are grateful to our families for their constant support and encouragement throughout this study.
Weiming Yan, Qiurui He and Lin Xiao are Co-first author.
Funding
This work was supported by the grants from the National Natural Science Foundation of China (Grant number: 82301245), the Natural Science Foundation of Fujian Province, China (Grant number: 2024J011148), the Joint Funds for the innovation of science and Technology, Fujian province (Grant number: 2024Y9653), the Postdoctoral Science Foundation of the Fuzhou General Hospital (Grant number: 48678) and the Natural Science Foundation of Zhangzhou City, China (Grant number: ZZ2024J57).
Disclosure
The authors declared no potential conflicts of interest regarding the research, authorship, or publication of this article.
References
1. Kumar S, Islam R, O’Connor W, et al. A metabolomic analysis on the toxicological effects of the universal solvent, dimethyl sulfoxide. Comp Biochem Physiol C Toxicol Pharmacol. 2025;287:110073. doi:10.1016/j.cbpc.2024.110073
2. Kligman AM. Dimethyl Sulfoxide. 2. JAMA. 1965;193:923–928. doi:10.1001/jama.1965.03090110061015
3. Kaczor-Kaminska M, Kaszuba K, Bilska-Wilkosz A, et al. Dimethyl Sulfoxide (DMSO) as a potential source of interference in research related to sulfur metabolism-a preliminary study. Antioxidants. 2024;13(5):582. doi:10.3390/antiox13050582
4. Sasatani M, Xi Y, Daino K, et al. Rev1 overexpression accelerates N -methyl- N -nitrosourea (MNU)-induced thymic lymphoma by increasing mutagenesis. Cancer Sci. 2024;115(6):1808–1819. doi:10.1111/cas.16159
5. Kong Q, Han X, Cheng H, et al. Lycium barbarum glycopeptide (wolfberry extract) slows N-methyl-N-nitrosourea-induced degradation of photoreceptors. Neural Regen Res. 2024;19(10):2290–2298. doi:10.4103/1673-5374.390958
6. Tawarayama H, Uchida K, Hasegawa H, et al. Estrogen, via ESR2 receptor, prevents oxidative stress-induced Muller cell death and stimulates FGF2 production independently of NRF2, attenuating retinal degeneration. Exp Eye Res. 2024;248:110103. doi:10.1016/j.exer.2024.110103
7. Zhu Y, Zhao X, Japhet TJ, Chen J, Zhao Q. Sampling of N-methyl-N-nitrosourea-induced rat’s retina damage and multi-index evaluation of pathological changes. J Vis Exp. 2024;(211).
8. Delgado-Goni T, Martín‐Sitjar J, Simões RV, et al. Dimethyl sulfoxide (DMSO) as a potential contrast agent for brain tumors. NMR Biomed. 2013;26(2):173–184. doi:10.1002/nbm.2832
9. Rosch S, Werner C, Müller F, et al. Photoreceptor degeneration by intravitreal injection of N-methyl-N-nitrosourea (MNU) in rabbits: a pilot study. Graefes Arch Clin Exp Ophthalmol. 2017;255(2):317–331. doi:10.1007/s00417-016-3531-7
10. Yan W, He Q, Long P, et al. A novelly-spatiotemporal characterization of the disease course in the MNU-induced retinitis pigmentosa model. J Inflamm Res. 2024;17:9243–9254. doi:10.2147/JIR.S474102
11. Tanaka M, Abe S. Augmented anti-tumour effect of lipopolysaccharide with G-CSF without enhancing body weight loss in mice bearing MH134 hepatoma. Eur J Pharmacol. 2022;934:175206. doi:10.1016/j.ejphar.2022.175206
12. Zhan J, Ma X, Liu D, et al. Gut microbiome alterations induced by tributyltin exposure are associated with increased body weight, impaired glucose and insulin homeostasis and endocrine disruption in mice. Environ Pollut. 2020;266(Pt 3):115276. doi:10.1016/j.envpol.2020.115276
13. Jarrar Q, Ayoub R, Jarrar Y, et al. Flumazenil pretreatment reduces mefenamic acid-induced central nervous system toxicity in mice. J Integr Neurosci. 2023;22(4):104. doi:10.31083/j.jin2204104
14. Mohamad Shalan NAA, Mustapha NM, Mohamed S. Chronic toxicity evaluation of Morinda citrifolia fruit and leaf in mice. Regul Toxicol Pharmacol. 2017;83:46–53. doi:10.1016/j.yrtph.2016.11.022
15. Nguyen KC, Zhang Y, Todd J, et al. Biodistribution and systemic effects in mice following intravenous administration of cadmium telluride quantum dot nanoparticles. Chem Res Toxicol. 2019;32(8):1491–1503. doi:10.1021/acs.chemrestox.8b00397
16. Chadalavada D, Adamson TW, Burnett JC, et al. Irradiated compared with nonirradiated NSG mice for the development of a human B-cell lymphoma model. Comp Med. 2014;64(3):179–185.
17. Suzuki H, Okada Y. Comparative toxicity of dinophysistoxin-1 and okadaic acid in mice. J Vet Med Sci. 2018;80(4):616–619. doi:10.1292/jvms.17-0377
18. Uttekar PS, Yadav VD, Bhagwat DA. 1, 2-Dihexadecanoyl-sn-glycero-3-phosphoethanolamin (DPPE), doxorubicin and folic acid conjugated micelles for cancer management in tumor bearing BALB/c mice. Bioorg Med Chem Lett. 2021;50:128337. doi:10.1016/j.bmcl.2021.128337
19. Dwivedi AK, Mallawaarachchi I, Alvarado LA. Analysis of small sample size studies using nonparametric bootstrap test with pooled resampling method. Stat Med. 2017;36(14):2187–2205. doi:10.1002/sim.7263
20. Neuhauser M, Jockel K. A bootstrap test for the analysis of microarray experiments with a very small number of replications. Appl Bioinformatics. 2006;5(3):173–179. doi:10.2165/00822942-200605030-00005
21. Swiecilo A, Januś E, Krzepiłko A, et al. The effect of DMSO on Saccharomyces cerevisiae yeast with different energy metabolism and antioxidant status. Sci Rep. 2024;14(1):21974. doi:10.1038/s41598-024-72400-4
22. Kim K, Lee S. Combined toxicity of dimethyl sulfoxide (DMSO) and vanadium towards zebrafish embryos (Danio rerio): unexpected synergistic effect by DMSO. Chemosphere. 2021;270:129405. doi:10.1016/j.chemosphere.2020.129405
23. Haschek WM, Baer KE, Rutherford JE. Effects of dimethyl sulfoxide (DMSO) on pulmonary fibrosis in rats and mice. Toxicology. 1989;54(2):197–205. doi:10.1016/0300-483X(89)90045-0
24. Huang Y, Cartlidge R, Walpitagama M, et al. Unsuitable use of DMSO for assessing behavioral endpoints in aquatic model species. Sci Total Environ. 2018;615:107–114. doi:10.1016/j.scitotenv.2017.09.260
25. Fry LJ, Querol S, Gomez SG, et al. Assessing the toxic effects of DMSO on cord blood to determine exposure time limits and the optimum concentration for cryopreservation. Vox Sang. 2015;109(2):181–190. doi:10.1111/vox.12267
26. Da Silva Franchi CA, Bacchi MM, Padovani CR, et al. Thymic lymphomas in Wistar rats exposed to N-methyl-N-nitrosourea (MNU). Cancer Sci. 2003;94(3):240–243. doi:10.1111/j.1349-7006.2003.tb01427.x
27. Guo D, Sun Y, Wu J, et al. Photoreceptor-targeted extracellular vesicles-mediated delivery of Cul7 siRNA for retinal degeneration therapy. Theranostics. 2024;14(13):4916–4932. doi:10.7150/thno.99484
28. Song P, Li X, Chen S, et al. YTHDF1 mediates N -methyl- N -nitrosourea-induced gastric carcinogenesis by controlling HSPH1 translation. Cell Prolif. 2024;57(7):e13619. doi:10.1111/cpr.13619
 © 2025 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 4.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2025 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 4.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.